Org. Biomol. Chem., 2011, 9, 6895-6898.
-
71. Palladium-catalyzed C–H acetoxylation of 2-methoxyimino-2-aryl-acetates and acetamides
Wang, L. Xia, X.-D. Guo, W. Chen, J.-R.* Xiao, W.-J.*
Org. Biomol. Chem., 2011, 9, 6895-6898.
DOI: 10.1039/C1OB05887H
Org. Biomol. Chem., 2011, 9, 5280-5287.
-
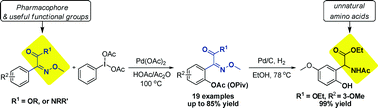
70. Pyrrolidinyl-sulfamide derivatives as a new class of bifunctional organocatalysts for direct asymmetric Michael addition of cyclohexanone to nitroalkenes
Chen, J.-R.* Fu, L. Zou, Y.-Q. Chang, N.-J. Rong, J. Xiao, W.-J.*
Org. Biomol. Chem., 2011, 9, 5280-5287.
DOI: 10.1039/C1OB05442B
Chem. Commun., 2011, 47, 8337-8339.
-
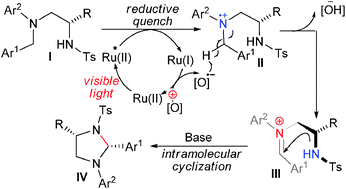
69. Visible light-induced intramolecular cyclization reactions of diamines: a new strategy to construct tetrahydroimidazoles
Xuan, J. Cheng, Y. An, J. Lu, L.-Q. Zhang, X.-X. Xiao, W.-J.*
Chem. Commun., 2011, 47, 8337-8339.
DOI: 10.1039/C1CC12203G
Angew. Chem. Int. Ed. 2012, 50, 7171-7175.
-
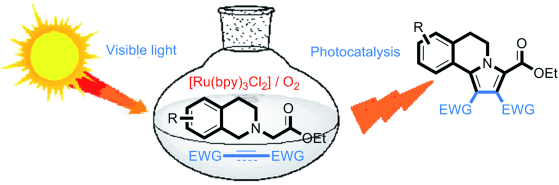
68.Visible-Light-Induced Oxidation/[3+2] Cycloaddition/Oxidative Aromatization Sequence: A Photocatalytic Strategy To Construct Pyrrolo[2,1-a]isoquinolines
Zou, Y.-Q. Lu, L.-Q. Fu, L. Chang, N.-J. Rong, J. Chen, J.-R.* Xiao, W.-J.*
Angew. Chem. Int. Ed. 2012, 50, 7171-7175.
DOI: 10.1002/anie.201102306
Synthesis, 2011, 12, 1847-1852.
-
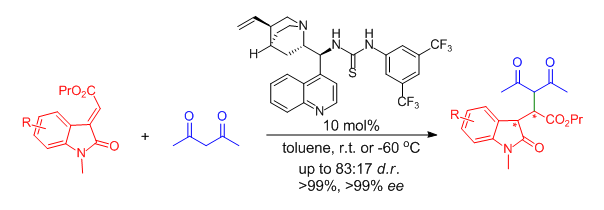
67.Organocatalytic Conjugate Additions of Acetylacetone to 3-Ylideneoxindoles: A Direct Access to Highly Enantioenriched Oxindole Derivatives
Duan, S.-W. Lu, H.-H. Zhang, F.-G. Xuan, J. Chen, J.-R.* Xiao, W.-J.*
Synthesis, 2011, 12, 1847-1852.
DOI: 10.1055/s-0030-1260463
Chemistry&ndashAn European Journal., 2011, 17, 6484-6491.
-
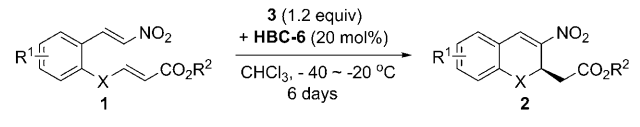
66.Enantioselective Intramolecular Crossed Rauhut–Currier Reactions through Cooperative Nucleophilic Activation and Hydrogen-Bonding Catalysis: Scope and Mechanistic Insight
Wang, X.-F. Peng, L. An, J. Li, C. Yang, Q.-Q. Lu, L.-Q. Gu, F.-L.* Xiao, W.-J.*
Chemistry&ndashAn European Journal., 2011, 17, 6484-6491.
DOI: 10.1002/chem.201100479
Synlett,2011, 1000-1004.
-
65.Phosphine-Catalyzed [3+2] Cycloadditions of 2-Phenyl-4-Arylidene-5(4H)-
Zou, Y.-Q. Li, C. Rong, J. Hao, Y. Chen, J.-R.* Xiao, W.-J.*
Synlett,2011, 1000-1004.
DOI: 10.1055/s-0030-1259709
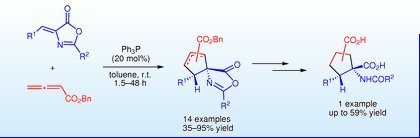
Org. Lett., 2011, 13, 2290-2293.
-
64.Facile Synthesis of Enantioenriched Cγ-Tetrasubstituted -Amino Acid Derivatives via an Asymmetric Nucleophilic Addition/Protonation Cascade
Duan, S.-W. An, J. Chen, J.-R.* Xiao, W.-J.*
Org. Lett., 2011, 13, 2290-2293.
DOI: 10.1021/ol200550y
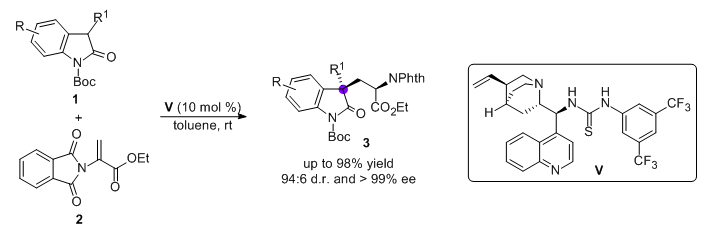
Adv. Synth. Catal., 2011, 353, 617-623.
-
63. Organocatalytic Multiple Cascade Reactions: A New Strategy for the Construction of Enantioenriched Tetrahydrocarbazoles
Cao, Y.-J. Cheng, H.-G. Lu, L.-Q. Zhang, J.-J. Cheng, Y. Chen, J.-R.* Xiao, W.-J.*
Adv. Synth. Catal., 2011, 353, 617-623.
DOI: 10.1002/adsc.201000610
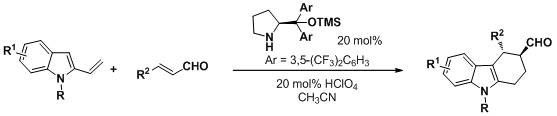
Org. Lett., 2011, 13, 808-811.
-
62. Catalytic Asymmetric Aza-Michael−Michael Addition Cascade: Enantioselective Synthesis of Polysubstituted 4-Aminobenzopyrans
Wang, X.-F. An, J. Zhang, X.-X. Tan, F. Chen, J.-R.* Xiao, W.-J.*
Org. Lett., 2011, 13, 808-811.
DOI: 10.1021/ol1031188

ChemCatChem 2011, 3, 679-683.
-
61.A Practical and Enantioselective Approach to Tetrahydrocarbazoles by Asymmetric Organocatalysis
Zhu, X.-Y. An, X.-L. Li, C.-F. Zhang, F.-G. Hua, Q.-L. Chen, J.-R.* Xiao, W.-J.*
ChemCatChem 2011, 3, 679-683.
DOI: 10.1002/cctc.201000379
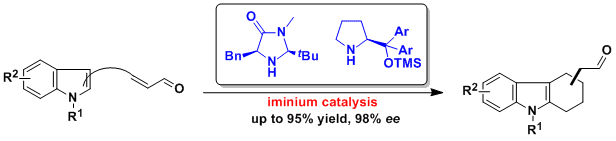
Chem. Commun. 2011, 47, 1869-1871.
-
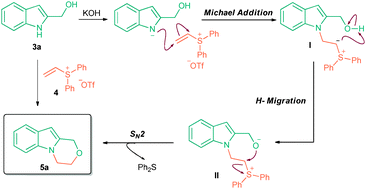
60.Efficient and general synthesis of oxazino[4,3-a]indoles by cascade addition-cyclization reactions of (1H-indol-2-yl)methanols and vinyl sulfonium salts
An, J. Chang, N.-J. Song, L.-D. Jin, Y.-Q. Ma, Y. Chen, J.-R.* Xiao, W.-J.*
Chem. Commun. 2011, 47, 1869-1871.
DOI: 10.1039/C0CC03823G
J. Org. Chem., 2011, 76, 281-284.
-
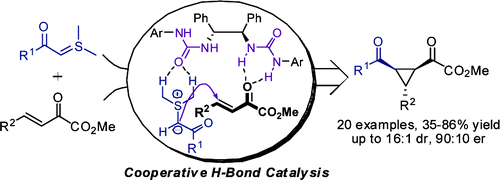
59.Asymmetric Cyclopropanation of β,γ-Unsaturated α-Ketoesters with Stabilized Sulfur Ylides Catalyzed by C2-Symmetric Ureas
Cheng, Y. An, J. Lu, L.-Q. Luo, L. Wang, Z.-Y. Chen, J.-R.* Xiao, W.-J.*
J. Org. Chem., 2011, 76, 281-284.
DOI: 10.1021/jo101699r
Tetrahedron2011, 67, 446-451.
-
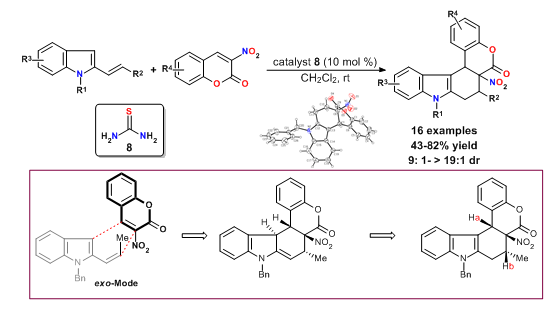
58. Brønsted acid catalyzed DielseAlder reactions of 2-vinylindoles and 3-nitrocoumarins: an expedient synthesis of coumarin-fused Tetrahydrocarbazoles
Tan, F. Li, F. Zhang, X.-X. Wang, X.-F. Cheng, H.-G. Chen, J.-R.* Xiao, W.-J.*
Tetrahedron2011, 67, 446-451.
DOI: 10.1016/j.tet.2010.11.019